The Physics Breakthrough
Accelbeam solved the penetration depth problem that has kept microwave chemistry at bench scale for decades.
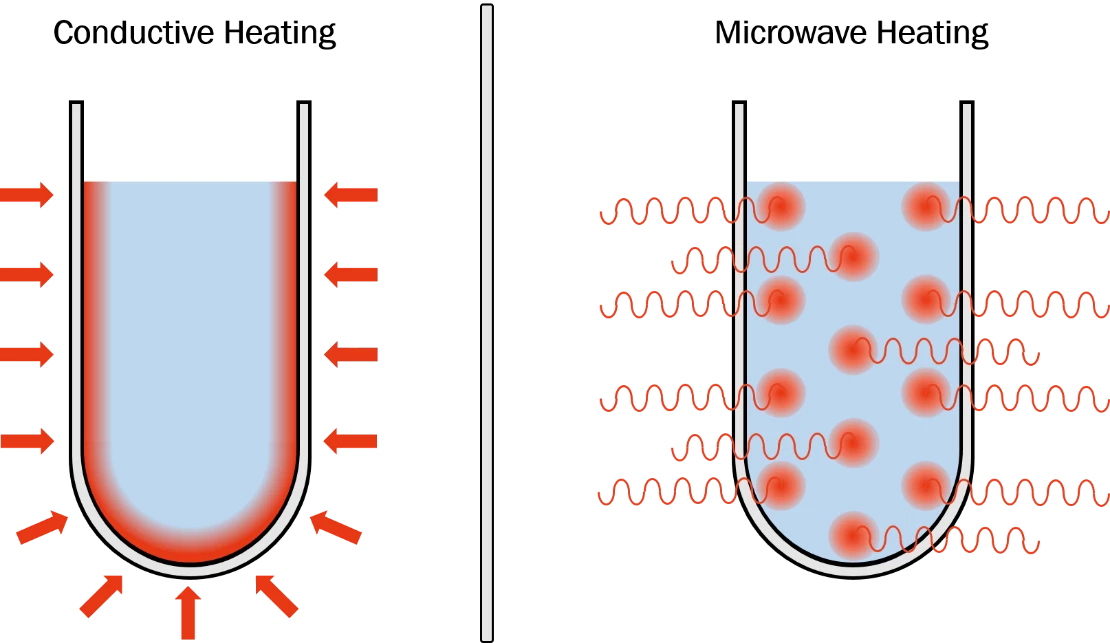
Volumetric Microwave Heating
Unlike conventional heating that heats vessel walls and waits for conduction, Accelbeam's CST delivers microwave energy directly to the entire reaction volume. 95% power uniformity at scale - vs. 40% for current microwave reactors.
Linear Scale-Up
Reaction kinetics remain identical regardless of volume. What works in a 100mL CEM vial works identically at 50L+ in Accelbeam's reactor. No reformulation required. Direct bench-to-commercial in the same technology.
Modular Reactor Stacks
Scale output by adding reactor stacks - not building bigger reactors or new plants. Deployable in weeks, not years. Enables distributed, rapid-response manufacturing.